TLDR
- Lunai Bioworks stock jumps 33.69% after pancreatic cancer breakthrough.
- Lunai Bioworks’ immune therapy shows tumor regression in pancreatic cancer models.
- LNAI achieves a major breakthrough in pancreatic cancer treatment, boosting stock.
- Lunai Bioworks’ immune therapy shows promise, pushing stock up by 33.69%.
- Stock for Lunai Bioworks rises 33.69% following breakthrough in cancer therapy.
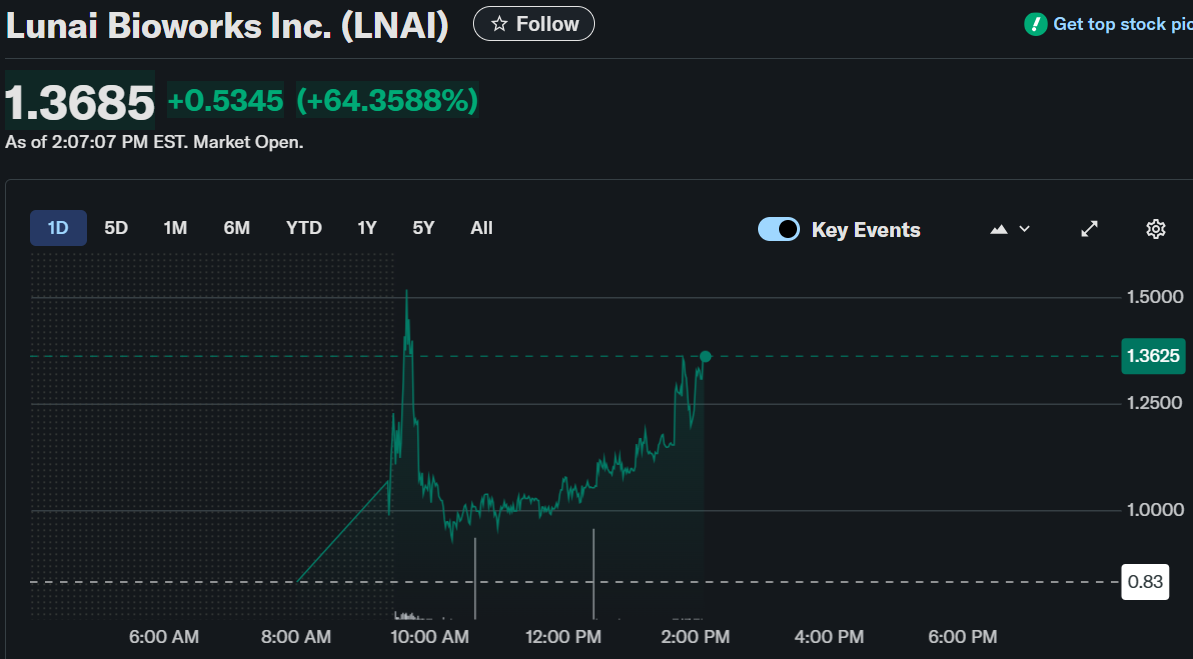
Lunai Bioworks (LNAI) experienced a significant surge in stock price, rising by 33.69% to $1.1150 during market hours.
Lunai Bioworks Announces Breakthrough in Pancreatic Cancer Therapy
LNAI has achieved a critical milestone in its fight against pancreatic cancer. The company revealed that its next-generation immune cell therapy resulted in the complete regression of both primary and metastatic pancreatic tumors in humanized mouse models. This breakthrough is the focus of a recent Brief Report published in the scientific journal Vaccines. The publication, released on November 2, 2025, provides detailed findings of Lunai’s second-generation clinical-grade dendritic cell (DC) therapy.
Pancreatic cancer is notoriously difficult to treat and remains one of the deadliest cancers. Conventional therapies often fail due to the cancer’s resistance to treatment and late-stage diagnoses. Lunai’s new therapy, which utilizes genetically engineered dendritic cells derived from stem cells, is designed to activate the immune system to combat cancer. This approach is a significant advancement in immunotherapy, showing promise not only in pancreatic cancer but potentially in a wide range of solid tumors.
New Clinical-Grade Therapy Shows Potent Results in Preclinical Models
Lunai’s second-generation DC therapy uses an optimized, clinically compliant construct that retains the same efficacy demonstrated in earlier studies. The new construct features an enhanced mammalian promoter and genetic elements aimed at improving therapeutic outcomes. In preclinical models, the therapy triggered a robust immune response, activating cytotoxic T cells and NK cells. This led to the complete regression of both primary and metastatic pancreatic tumors in the models.
The study’s findings are a direct continuation of Lunai’s previous work, which demonstrated the effectiveness of its research-grade DC product in early trials. The latest research has refined the therapy to meet clinical standards, making it closer to human clinical trials. This advancement moves Lunai Bioworks from research and development into the clinical translation phase, a crucial step for the company as it aims to bring its innovative therapy to market.
Lunai Bioworks Positioned at the Forefront of Immunotherapy Innovation
David Weinstein, CEO of Lunai Bioworks, emphasized the importance of the company’s progress. He stated that the optimization of their vector platform brings them closer to initiating IND-enabling studies. According to Weinstein, this breakthrough represents a significant step forward in Lunai’s journey from laboratory innovation to clinical applications. The company is now preparing to advance its allogeneic DC platform to potential clinical trials.
This achievement marks a major inflection point for Lunai Bioworks, which continues to innovate within the biopharmaceutical space. As the company moves toward clinical trials, it will work on partnering with organizations to further develop and commercialize its therapies. Lunai Bioworks remains committed to revolutionizing cancer treatment, positioning itself as a leader in next-generation immune-based therapies.